Contact us
Need more information? Your local key account manager will be in contact shortly.
This page is intended for UK healthcare professionals and other relevant decision makers only. If you are a member of the public, please click here.
This portal is funded and owned by Novartis Pharmaceuticals UK Ltd and includes content approved by Novartis.
Adverse events reporting information can be found in the footer of this page.
This page is intended for UK healthcare professionals currently prescribing KESIMPTA®▼ (ofatumumab), who are interested in learning more about the KesimptaConnect Programme.
KESIMPTA is indicated for the treatment of adult patients with relapsing forms of multiple sclerosis with active disease defined by clinical or imaging features.1
KesimptaConnect has been developed and funded by Novartis Pharmaceuticals UK Limited. KesimptaConnect pre-initiation, nurse service and home delivery is offered as a package deal. KesimptaConnect portal is offered as a patient support programme. KesimptaConnect is for patients who are receiving KESIMPTA or for whom the prescribing decision has been made. A service-level agreement is required for each NHS organisation participating.
5369 active patients, as of November 2024.2
83 sites signed up.3
98% of surveyed patients (N=1003) are extremely satisfied with the KesimptaConnect Nurse service overall.4
KesimptaConnect offers dedicated, personalised support to patients and healthcare professionals throughout the KESIMPTA journey.
Enrollment provides your patients unique access to a wide range of services, including injection support, home deliveries, tailored information and interactive content covering wider aspects of living with RMS, as well as a dedicated, experienced nurse support team. The programme has been designed to offer personalised support in conjunction with the medical care provided by your hospital’s MS team.
Healthcare professionals are also able to access a personalised support service based on the needs of their centre.
1. KesimptaConnect Homecare: in line with the National Homecare Medicines Committee (NHMC) Pharma-funded Homecare Services guidelines, Novartis has commissioned Lloyds Pharmacy Clinical Homecare Limited, Pharmaxo Pharmacy Services Limited and Sciensus Limited to provide a homecare service for patients prescribed KESIMPTA.
2. KesimptaConnect Nurse services: nurses who can provide virtual or face-to-face appointments with your patients to provide injection guidance and support in the first year of each patient’s treatment journey.
3. The KesimptaConnect app: provides a hub of information and resources to your patient, including an injection tracker to support treatment adherence.
There is no obligation to use all elements of this programme; it is designed to allow you to choose the components that best suit the needs of your patient.
Discuss with your local Novartis representative which elements are right for your patients and the MS services you provide.
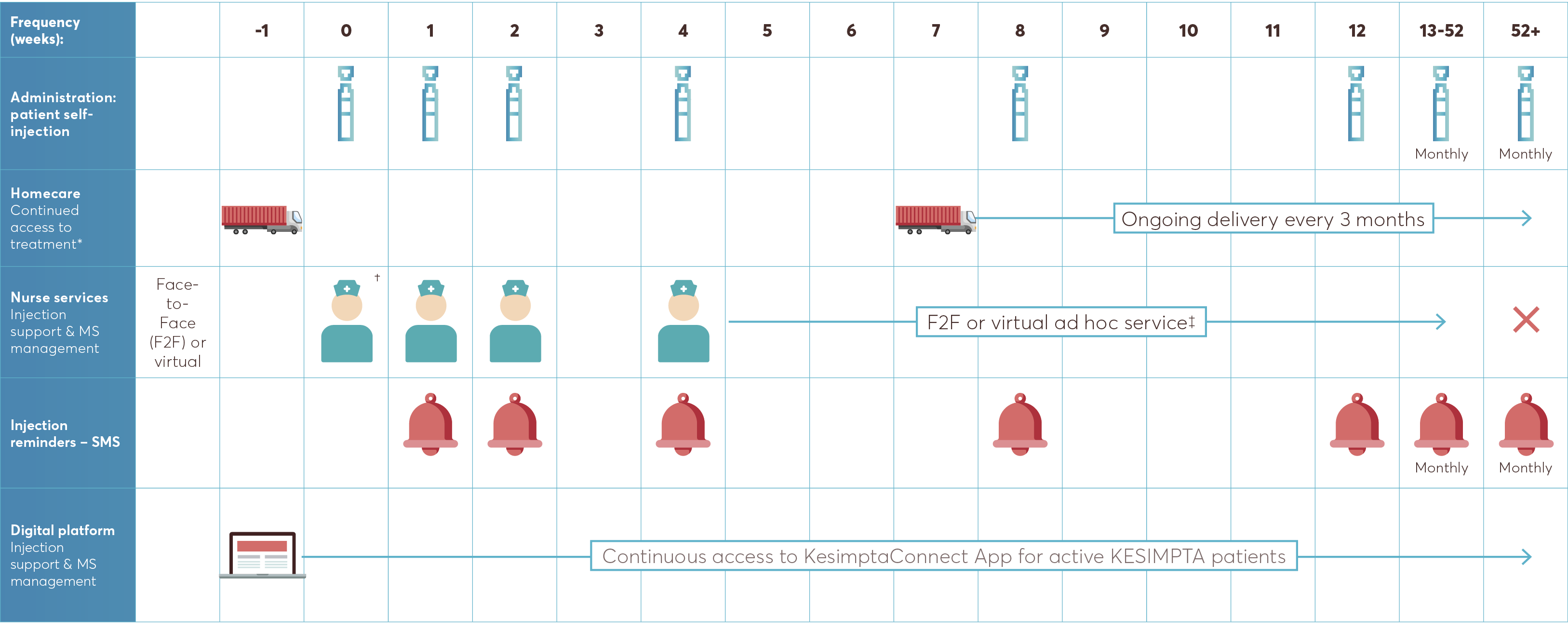
As part of KesimptaConnect, your patients have access to a homecare service:
The service will deliver the KESIMPTA pens straight to your patient’s door at a time and date that is convenient to them. Delivery schedule: one delivery to cover the first 4 injections and from Week 8, delivery every 3 months.†
The Homecare suppliers have a robust prescription management system. They will proactively seek any missing prescriptions in order to ensure your patients receive their KESIMPTA at the correct time.
†For ongoing deliveries from Week 8, if you or your patient prefer monthly delivery, this is also an option.
The KesimptaConnect Nurse team has experience caring for a wide range of patients with MS and have in-depth knowledge of KESIMPTA.
Once KESIMPTA has been prescribed, the nurse team can provide self-injection support, designed to ensure that your patient is both:
They will also keep you informed of your patient’s progress.
Each KesimptaConnect Nurse can support your patients with health psychology approaches.
The KesimptaConnect Nurse will provide support using a self-injection demonstration kit and answer any further questions your patient may have
At the first appointment, the KesimptaConnect Nurse will conduct personalisation screening, to understand the patient’s support needs, agree a contact schedule with the patient and arrange the next call or visit
The KesimptaConnect Nurse will make contact with the patient ahead of each scheduled injection for the first 12 weeks to check on confidence with self-injection and provide personalised support
Subsequent interactions will be determined by the patient and KesimptaConnect Nurse (as required) for up to 12 months
Visit summaries will be sent to you by the KesimptaConnect Nurse after each patient interaction to keep you informed of your patient’s progress
Injection reminders can be set up by the KesimptaConnect Nurse so that the patient is sent SMS reminders one day before and on the day their injection is due. This option will be discussed with the patient at the first appointment
The service will ensure that your patient is adequately supported until they are comfortable with their once-monthly self-administration.
Once your patient is comfortable, they will also be supported in accessing the designated KesimptaConnect App for further ongoing support.
For more information and details on how to enrol your patients in the KesimptaConnect programme, please speak to your local Novartis representative.
The KesimptaConnect app provides educational support and resources tailored to each patient, including:
Information on KESIMPTA to empower your patient to understand their MS and provide tools to help them manage it
MS management support to provide articles and interactive content on wider aspects of living with MS (e.g., managing fatigue, emotional wellbeing, keeping active, work)
Injection reminders and tracking will enable your patients to track when to take their injection, record when they took injections and which injection site was used
The KesimptaConnect nurse will support the patient in accessing the app, which is available on both mobile phone and web browsers.
Should you wish to find out more about the app please reach out to your local Novartis representative.
KesimptaConnect checklist | |
Set up | Sign the Overview and Offer Form with Novartis Pharmaceuticals UK Ltd |
Sign the Programme Authorisation Document with IQVIA IES UK Ltd | |
Sign the Service Level Agreement with your Homecare partner of choice | |
Once KESIMPTA has been chosen as the treatment of choice | |
Make your patient aware of the KesimptaConnect service and choose the elements that would be best for your patient | |
Homecare | Register your patient for KesimptaConnect by contacting the relevant Homecare company |
Injection support and nurse services | Register your patient for KesimptaConnect injection support and nurse services by emailing the completed patient registration form to [email protected] |
KesimptaConnect App | Register your patient for the KesimptaConnect App by emailing the completed patient registration form to [email protected] |
View the KesimptaConnect touchpoints below
*Homecare: initial phase – one delivery for first 4 injections, ongoing – delivery every 3 months after initiation. For ongoing, if you or your patient prefer monthly delivery, this is also an option.
†The first injection should be performed under the guidance of the KesimptaConnect Nurse or appropriately trained HCP.
‡As required by the patient. Nurse completes assessment questionnaire to determine the level of support the patient needs.
MS, multiple sclerosis; NHS, National Health Service; NHMC, national homecare medicines committee; RMS, relapsing forms of multiple sclerosis.
References
KESIMPTA (ofatumumab) Summary of Product Characteristics.
Novartis Data on File, OFA231, 2025.
Novartis Data on File, OFA019, 2024.
Novartis Data on File, OFA017, 2024.
UK | March 2025 | FA-11213993
Adverse events should be reported. Reporting forms and information can be found at www.mhra.gov.uk/yellowcard. Adverse events should also be reported to Novartis online through the pharmacovigilance intake (PVI) tool at www.novartis.com/report, or alternatively email [email protected] or call 01276 698370.